ULTRANANOTECH, Uncategorized
MXenes-2D Materials
MXenes-2D Materials:
MXenes – pronounced ‘max-eens’ – first discovered in 2011, are ceramics that comprise one of the largest families of two-dimensional (2D) materials.
MXenes are made from a bulk crystal called MAX. Unlike most 2D ceramics, MXenes have inherently good conductivity and excellent volumetric capacitance because they are molecular sheets made from the carbides and nitrides of transition metals like titanium. MXenes have already found applications ranging from energy storage to medicine and optoelectronics.
What makes MXenes so interesting is the fact that this material class could conceivably consist of any of millions of possible arrangements of transition metals (like molybdenum or titanium), carbon, and nitrogen.
By using a high-throughput computational platform and scanning through the formation energies of millions of alloying configurations, researchers estimate that there are more than a million of stable MXene compounds remaining to be discovered.
MAX Phases:
There is a large family of ternary carbides (ternary is an adjective meaning ‘composed of three’) with the general formula Mn+1AXn, where n = 1ñ3, M denotes a transition metal, A is an element such as aluminum or silicon, and X is either carbon or nitrogen. Researchers have termed these ductile and machineable ceramics MAX phases.
As a consequence of their layered structure, these materials kink and delaminate during deformation and also exhibit an unusual, and sometimes unique, combination of properties; they are not sure whether they want to be metals or ceramics. While they conduct heat and electricity like metals, they are elastically stiff, strong, brittle, and heat-tolerant like ceramics.
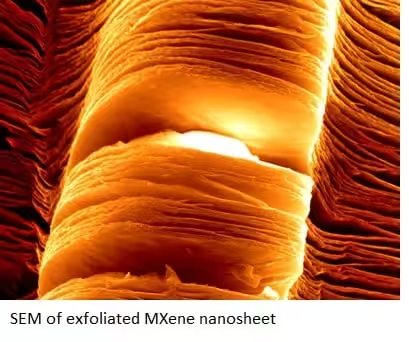
MXene Discoveries:
Two-dimensional (2D) structures, like graphene and molybdenum disulfide, are known to have unique properties. Therefore, having a new family of 2D structures with a wide range of chemistries can open the door for a better understanding of differences between properties of 2D and 3D materials, leading to the identification of useful properties of 2D carbides, nitrides, oxycarbides and other related structures, and finally result in new applications.
However, these ceramics have always been produced as three-dimensional materials, until researchers placed titanium-aluminum carbide (Ti3AlC2) powders in hydrofluoric acid at room temperature to selectively remove the aluminum. The result of this chemical process – referred to as exfoliation – essentially spreads out the layered carbide material and yields two-dimensional Ti3C2 nanosheets, which have since been coined MXene, as a kin to graphene.
MXene uses and applications:
MXene could be used in energy storage devices such as electrodes of Li-ion batteries, pseudo capacitors, etc. The researchers also envision its use as reinforcement in composites, similar to clays or graphene, which increase mechanical properties and decrease gas permeability of polymers. A variety of surface chemistries, presence of transition metal oxides and high surface area make MXene potentially attractive for catalytic applications.
Desalination and waste-water treatment:
The materials’ remarkable properties open new possibilities for MXenes in water desalination and wastewater treatment. This excitement comes from findings that Ti3C2 can trap the energy of sunlight to purify water by evaporation.
To investigate MXeneís possibilities in water purification, researchers fabricated a thin and flexible Ti3C2 membrane incorporating a polystyrene heat barrier to prevent the heat energy from escaping. This created a system that could float on water and evaporate some of the water with 84% efficiency at the illumination levels of natural sunlight.
Battery technology and energy storage
Computational studies have suggested that fully exfoliating or delaminating, certain MXenes would yield layers with exceptional charge capacities for use in battery anodes. In one report, scientists demonstrated successful intercalation of MXenes with several organic molecules, including dimethyl sulfoxide (DMSO), which allowed them to fully exfoliate stacked layers into MXene sheets and ultimately create MXene ‘paper’ by filtering flakes from the solution.
This flexible and electrically conductive paper showed a lithium-ion capacity of four times that of typical MXene material, with extremely high charging rates and a cyclability superior to graphite, which is used in commercial lithium-ion batteries.
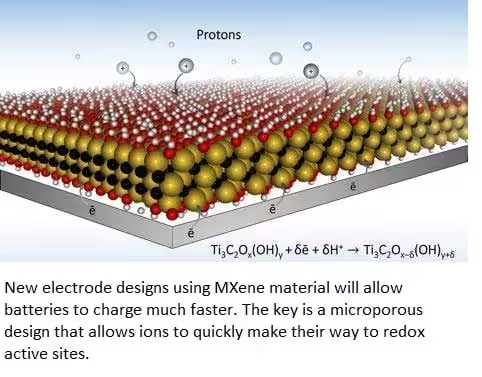
Researchers also have developed new electrode designs with MXene material, that will allow batteries to charge much faster.
Courtesy: ULTRANANOTECH PVT. LTD.
For more information visit us at: https://ultrananotec.com/

